Transformation of Energy
Overview-
This unit is designed to set the stage for the entire 8th grade year. The unit starts out with an in-depth investigation of mechanical energy, progresses to the topic of heat energy, and ends with an investigation into the energy carried by waves. Energy transfers and energy transformations as well as the construction of energy chains are a key focus of the unit
Big Ideas-
This unit is designed to set the stage for the entire 8th grade year. The unit starts out with an in-depth investigation of mechanical energy, progresses to the topic of heat energy, and ends with an investigation into the energy carried by waves. Energy transfers and energy transformations as well as the construction of energy chains are a key focus of the unit
Big Ideas-
- A clear definition of energy does not exist; therefore, we “define” energy by looking at its effects.· Energy can be categorized into many forms. These include kinetic, potential, heat, and electromagnetic energies.
- Energy can be transferred and/or transformed. In many cases the energy is transferred to particles that are too small for us to see.
- An energy chain can be used to diagram the flow of energy in a physical system.
- Temperature is a measure of the average kinetic energy of the particles in a substance. Heat energy is the total amount of random vibrational, kinetic energy of the particles in a substance. Heat energy and temperature are not synonyms.
- Heat energy can be transferred by conduction, by convention, and/or by radiation.
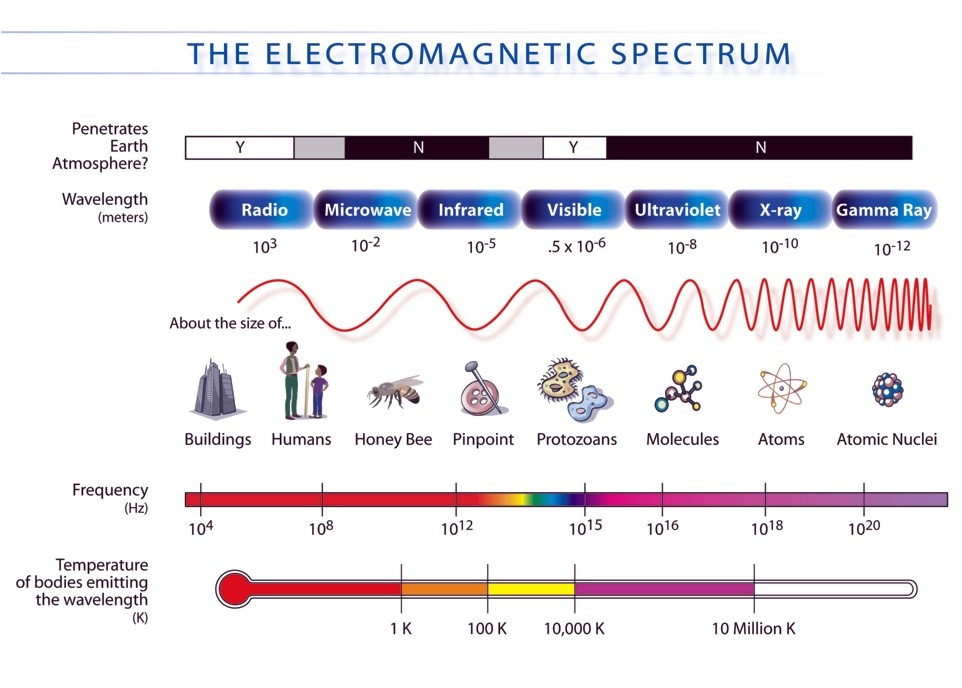
- Energy can be carried by the action of waves. Mechanical waves carry mechanical energy while electromagnetic waves carry electromagnetic energy.
- Waves transfer energy without transferring mass. The amount of energy transferred by a wave depends upon its amplitude, frequency, and wavelength.
- Different materials transfer energy at different rates.
(This information is taken from the Delaware Department of Education website under Science Education Unit Templates)